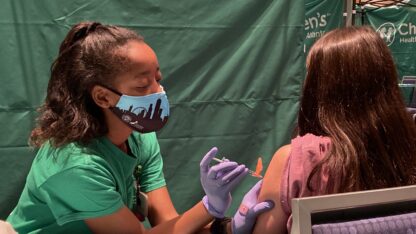
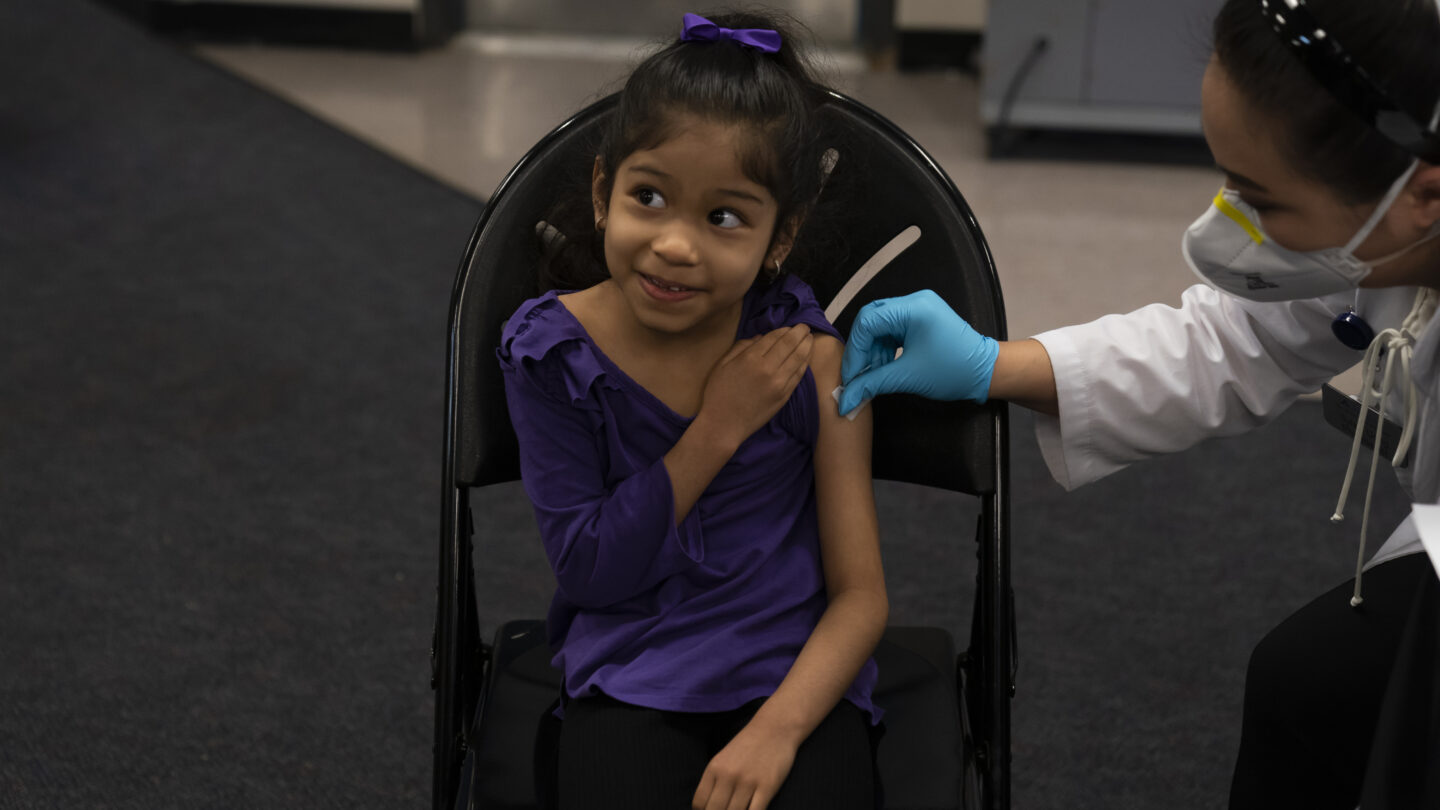
The Food and Drug Administration Tuesday authorized the first COVID-19 vaccine booster for children ages 5 to 11.
The authorization makes all children in that age group who received their second shot at least five months ago eligible to receive a third shot of the Pfizer-BioNTech vaccine.
The companies requested the authorization based on a small study that the companies and FDA said demonstrated a third shot is safe and can significantly boost antibody levels, countering waning immunity and providing added protection against the virus, including the more contagious omicron variant.
Until now, only children ages 12 and older and adults were eligible for a booster.
“While it has largely been the case that COVID-19 tends to be less severe in children than adults, the omicron wave has seen more kids getting sick with the disease and being hospitalized, and children may also experience longer-term effects, even following initially mild disease,” said FDA Commissioner Robert M. Califf in a statement.
“The FDA is authorizing the use of a single booster dose of the Pfizer-BioNTech COVID-19 Vaccine for children 5 through 11 years of age to provide continued protection against COVID-19,” Califf said. “Vaccination continues to be the most effective way to prevent COVID-19 and its severe consequences, and it is safe.”
It remains unclear how much of a demand there will be for the boosters. Less than a third of children ages 5 to 11 are fully vaccinated, and only about a quarter of adolescents have received a booster.
Many doctors and parents have been hoping for the authorization, saying it’s important to provide children with as much protection as possible, especially as the number of people catching the virus is rising again and so few people are now wearing masks.
“Since authorizing the vaccine for children down to 5 years of age in October 2021, emerging data suggest that vaccine effectiveness against COVID-19 wanes after the second dose of the vaccine in all authorized populations,” said Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, in a statement.
“The FDA has determined that the known and potential benefits of a single booster dose of the Pfizer-BioNTech COVID-19 vaccine for children 5 through 11 years of age at least five months after completing a primary series outweigh its known and potential risks and that a booster dose can help provide continued protection against COVID-19 in this and older age groups,” Marks said.
But others are skeptical, saying two shots are still providing strong protection against serious disease and death, and it remains unclear how much added protection a third shot will provide or how long it will last.
The FDA didn’t ask its outside advisers for input on the decision because the agency said the committee had previously discussed the issues and no new questions were raised.
The Centers for Disease Control and Prevention’s advisers on vaccine practices will meet Thursday and are expected to discuss implementation of a booster dose for this age group.
Copyright 2022 NPR. To see more, visit https://www.npr.org.
9(MDAxODM0MDY4MDEyMTY4NDA3MzI3YjkzMw004))
9(MDAxODM0MDY4MDEyMTY4NDA3MzI3YjkzMw004))